|
All rights reservedġ9 sp Hybridization Combination of one s and one p orbital. What is the shape of a carbon dioxide molecule? What are the bond angles? The shape of CO2 is linear. 
All rights reservedĮXERCISE! Draw the Lewis structure for CO2. To play movie you must be in Slide Show Mode PC Users: Please wait for content to load, then click to play Mac Users: CLICK HERE Copyright © Cengage Learning. All rights reservedġ5 An Orbital Energy-Level Diagram for sp2 Hybridizationġ6 The Hybridization of the s, px, and py Atomic Orbitalsġ7 Formation of C=C Double Bond in Ethylene All rights reservedġ4 Pi (Π) Bond Forms double and triple bonds by sharing electron pair(s) in the space above and below the σ bond. All rights reservedġ3 Sigma (Σ) Bond Electron pair is shared in an area centered on a line running between the atoms. Oriented perpendicular to the plane of the sp2 orbitals. Gives a trigonal planar arrangement of atomic orbitals. All rights reservedġ2 sp2 Hybridization Combination of one s and two p orbitals. All rights reservedġ1 Why can’t sp3 hybridization account for the ethylene molecule?ĬONCEPT CHECK! Why can’t sp3 hybridization account for the ethylene molecule? Because sp3 hybrid orbitals are at angles of 109.5o rather than the required 120o. All rights reservedġ0 Draw the Lewis structure for C2H4 (ethylene)?ĮXERCISE! Draw the Lewis structure for C2H4 (ethylene)? What is the shape of an ethylene molecule? What are the approximate bond angles around the carbon atoms? The shape of an ethylene molecule is trigonal planar around each carbon atom. All rights reservedħ An Energy-Level Diagram Showing the Formation of Four sp3 OrbitalsĬopyright © Cengage Learning. The four orbitals are identical in shape. Whenever a set of equivalent tetrahedral atomic orbitals is required by an atom, the localized electron model assumes that the atom adopts a set of sp3 orbitals the atom becomes sp3 hybridized. All rights reservedĦ sp3 Hybridization Combination of one s and three p orbitals. All rights reservedĥ Hybridization Mixing of the native atomic orbitals to form special orbitals for bonding. All rights reservedĤ Bonding in Methane Assume that the carbon atom has four equivalent atomic orbitals, arranged tetrahedrally. Because this would lead to two different types of C-H bonds and we know that methane has four identical C-H bonds that are 109.5° apart from each other (not 90° from each other). All rights reservedģ What is the valence electron configuration of a carbon atom?ĬONCEPT CHECK! What is the valence electron configuration of a carbon atom? Why can’t the bonding orbitals for methane be formed by an overlap of atomic orbitals? The valence electron configuration of a carbon atom is s2p2. What is the shape of a methane molecule? What are the bond angles? The shape of CH4 is tetrahedral. Presentation on theme: "Draw the Lewis structure for methane, CH4."- Presentation transcript:Ģ Draw the Lewis structure for methane, CH4.ĮXERCISE! Draw the Lewis structure for methane, CH4. en:User:Sinuhe Sinuhe ( en:User_talk:Sinuhe Talk). Legend: (cur) = this is the current file, (del) = delete this old version, (rev) = revert to this old version.Ĭlick on date to download the file or see the image uploaded on that date. (My feeble attempt at drawing methane in terms of structure) CC-BY-SA-3.0 Creative Commons Attribution-Share Alike 3.0 true true This licensing tag was added to this file as part of the GFDL licensing update. share alike – If you remix, transform, or build upon the material, you must distribute your contributions under the same or compatible license as the original.You may do so in any reasonable manner, but not in any way that suggests the licensor endorses you or your use. attribution – You must give appropriate credit, provide a link to the license, and indicate if changes were made.to share – to copy, distribute and transmit the work. 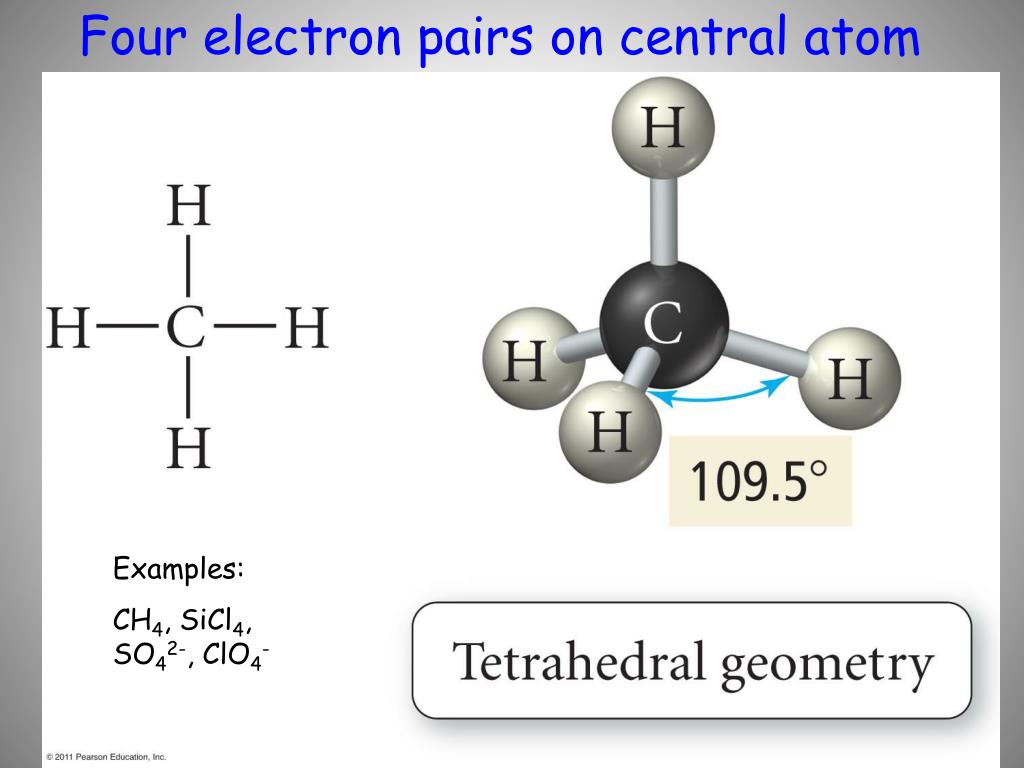 
This file is licensed under the Creative Commons Attribution-Share Alike 3.0 Unported license. 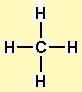
GFDL GNU Free Documentation License true true A copy of the license is included in the section entitled GNU Free Documentation License. Permission is granted to copy, distribute and/or modify this document under the terms of the GNU Free Documentation License, Version 1.2 or any later version published by the Free Software Foundation with no Invariant Sections, no Front-Cover Texts, and no Back-Cover Texts.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
